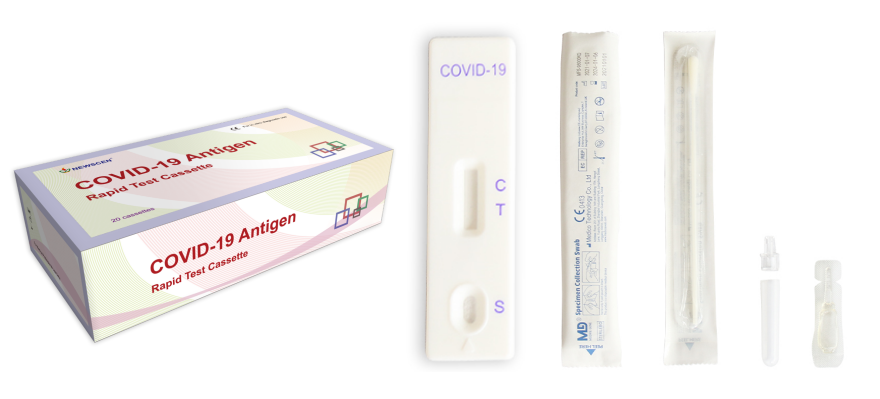
Key Words: Coronavirus (COVID-19) Antigen Rapid Test Cassette (Swab), COVID-19 Antigen Test Kit, SARS COV 2 Variant Test Kits
Sample type: Throat OR nasal swab
Detection type: Qualitative
Method: Colloidal Gold Rapid Test
Function: Diagnose
Certificate: ISO9001/ ISO13485/CE
Format: Strip, Cassette, Midstream
Sensitivity: 96.23%
Specificity: 99.11%
Accuracy: 98.56%
Reading time: 15 minutes
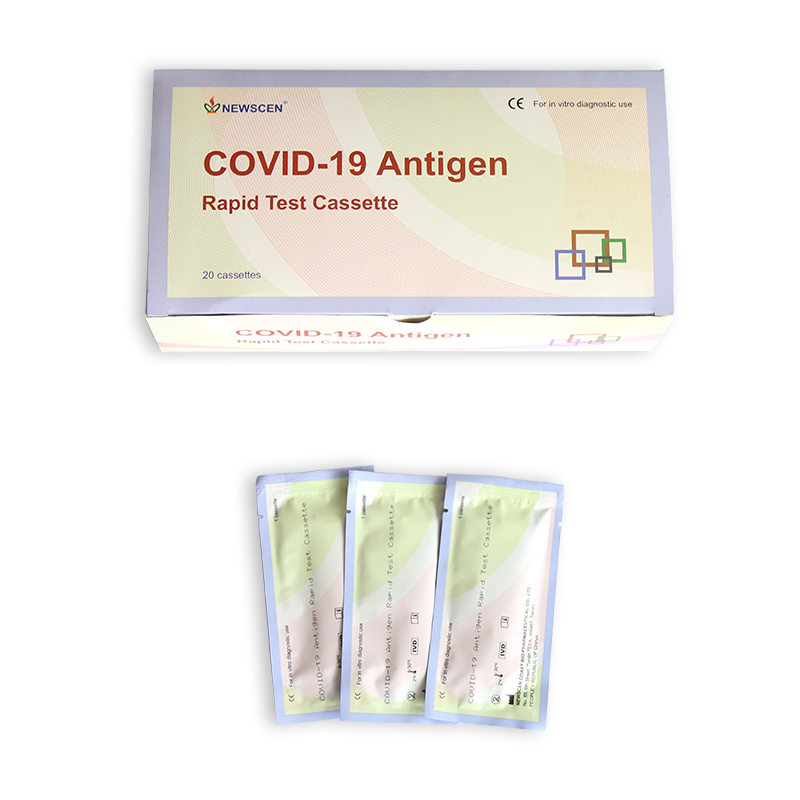
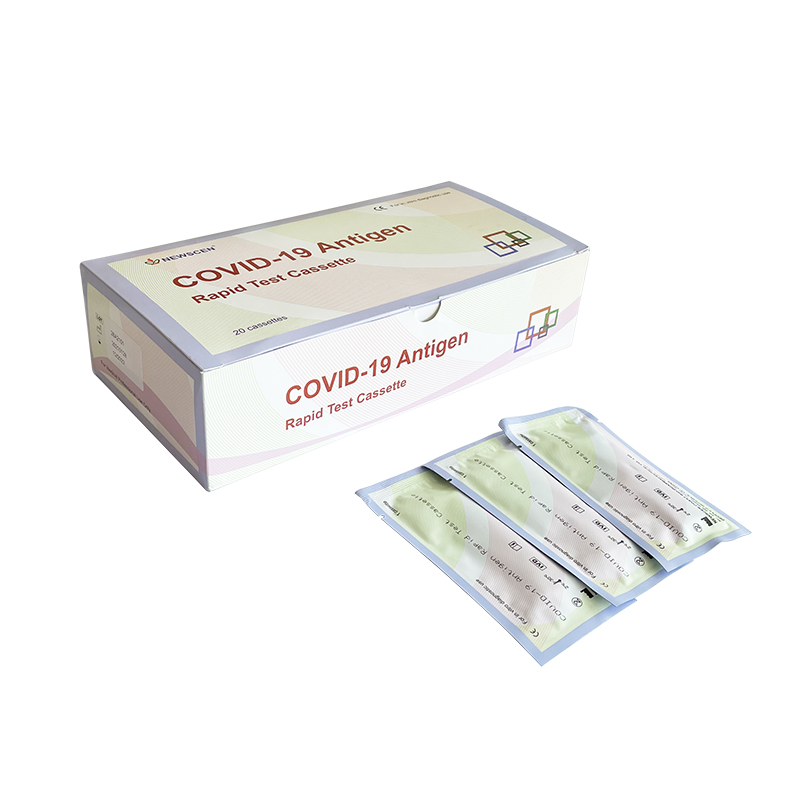
Packaging Details:
Pouch+Box+Carton packaging
(1) With our company’s Logo
(2) With the natural package
(3) With OEM package
(4) ODM
Description
1. Product Description:
COVID-19 Antigen Rapid Test Cassette is suitable for large-scale screening. It is an ideal alternative to RT-PCR for cases with suspected symptoms of COVID-19 within 5~7 days.
2. Intended Use:
COVID-19 Antigen Rapid Test Cassette is for in vitro qualitative detection of specific antigens to SARS-CoV-2 present in the human throat or nasal cavity. It cannot be used as the basis for the diagnosis and exclusion of COVID-19.
This reagent is used to detect cases with suspected symptoms of COVID-19 within 7 days. If suspected symptoms are more than 7 days, it is recommended to test with COVID-19 antibodies or nucleic acid reagents.
The main clinical symptoms of COVID-19 are: Fever, dry cough, fatigue, a few patients will have a stuffy nose, runny nose, and diarrhea.
3. Principle:
This kit uses a double-antibody sandwich immunoassay to detect specific antigens to SARS-CoV-2 in the human throat or nasal cavity. The membrane was precoated with SARS-CoV-2 specific antibody on the test zone and goat anti-mouse IgG antibody on the control zone. During the test, the specimen is allowed to react with SARS-CoV-2 specific antibody-colloidal gold particles conjugate, which was predried on the test. The conjugate binds to the SARS-CoV-2 forming an antibody~antigen complex. The complex moves forward on the membrane by the capillary action, then the complex is captured by a specific antibody to SARS-CoV-2 on the test zone to produce a visual red color line. The color of the line is positively correlated with the amount of SARS-CoV-2 in the specimen.
Regardless of the presence of SARS-CoV-2, as the mixture continues to move across the membrane to the control zone, the complex is captured by immobilized goat anti-mouse IgG antibodies to form a distinct red line.
4. Storage:
COVID-19 Antigen Rapid Test Cassette should be stored in a dark place at 2~30°C for 24 months from the date of manufacture. Keep the test cassette in a sealed pouch until use. Once you have taken the test cassette out of the pouch, use it immediately. Do not use the test beyond the indicated expiration date.
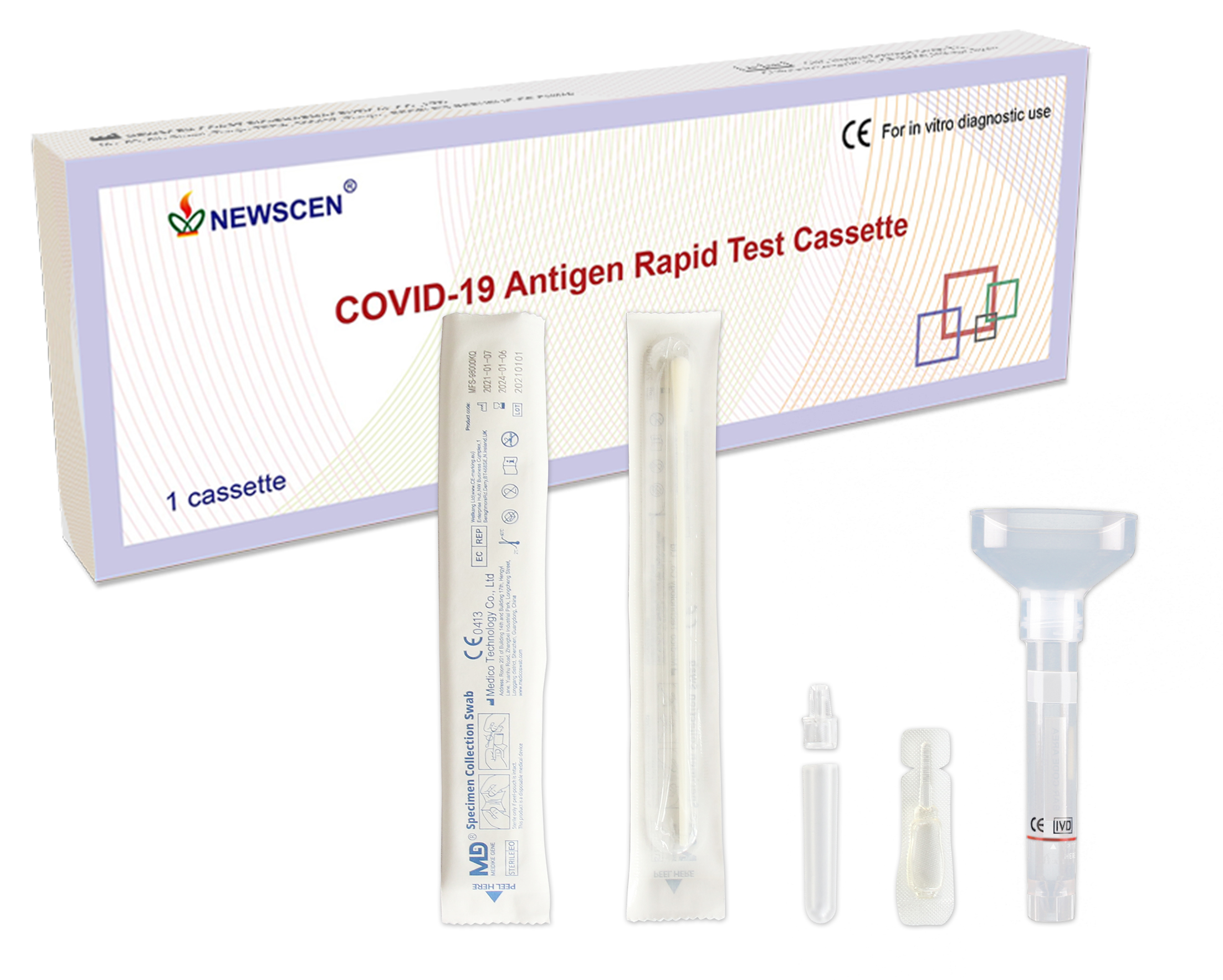
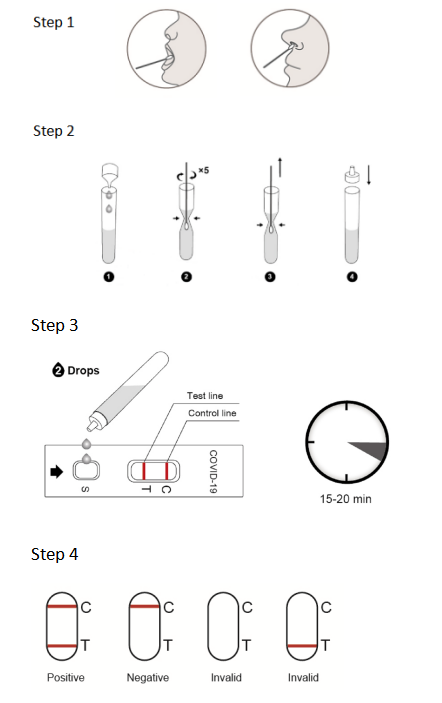
5. Procedure of NewScen COVID-19 Antigen Rapid Test Cassette:
5.1. All clinical samples must be at room temperature before beginning the assay.
5.2. Open the package, the pouch should be sealed well. If the test reagent store in the refrigerator, it should be restored to room temperature. Then open the pouch and take out the test cassette, place it on the platform.
5.3. Add 2 drops of patient sample extraction solution from the tube into S Well, Observe the result in 15~20 minutes, interpret the test result after 20 minutes may cause a false result.
6. Interpretation of Result:
6.1. Positive: One color line in the control zone (C) and one color line in the test zone (T). This indicates that the sample contains SARS-CoV-2 antigen.
6.2. Negative: Only one color line in the control zone (C). This indicates that no SARS-CoV-2 antigen has been detected.
6.3. Invalid: If no color line appears in the control zone (C), the test is invalid. Discard the test cassette and perform with a new cassette.
Built-In Control
COVID-19 Antigen Rapid Test Cassette has a built-in procedural control that demonstrates assay validity. A color line appeared on the control zone (C) indicates that the test runs correctly.
7. Limitation:
7.1. The kit is only used to detect human throat swabs or nasal swabs.
7.2. The accuracy of the test depends on the process of sample collection. Improper sample collection, improper sample storage, or repeated freezing and thawing of samples will affect the test results.
7.3. The test results of this reagent are for clinical reference only and should not be used as the sole basis for clinical diagnosis and treatment. The clinical management of the patient should be considered in combination with other laboratory tests of the patient’s symptoms/signs history and treatment response.
8.CE Qualified: