Key Words: SARS-CoV-2 (Covid-19) Diagnosis by IgG/IgM Rapid Test, COVID-19 Rapid Test Kit IgG + IgM (Colloidal Gold)
Material: Plastic
Usage/Application: Laboratory / Hospital / Pathology
Sample type: Blood/Serum/Plasma
Detection type:Qualitative
Method: Colloidal Gold Rapid Test
Function: Diagnose
Certificate: ISO9001/ ISO13485/CE
Format: Strip, Cassette, Midstream
Sensitivity: 96.79%
Specificity: 99.28%
Accuracy: 98.43%
Reading time: 10-20 minutes
Packaging Details:
Pouch+Box+Carton packaging
(1) With our company’s Logo
(2) With the natural package
(3) With OEM package
(4) ODM
Description
1.Product Description:
COVID-19 lgG/IgM Rapid Test Cassette is for the qualitative detection of COVID-19 IgG and IgM antibodies in human serum, plasma, or whole blood. It is suitable for screening a large population.
This reagent is CE marked and certified by China export white list.
2.Features of NewScen COVID-19 IgG/IgM Rapid Test Cassette:
- Serum, plasma, whole blood (fingertip/vein puncture ) supported
- Easy to sampling, effectively reduce the risk of infection in medical staffs
- Room temperature storage and transportation
- Easy-to-use, test result visually observed within 15 minutes
- Suitable for large scale screening
- Easier and faster to diagnose COVID-19
3.Principle:
This kit uses colloidal gold immunochromatography and indirect immune capture principle of comprehensive detection technology to detect IgG/IgM of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in human serum, plasma, and whole blood.
During detection, the colloidal gold-labeled recombinant antigens bind to the SARS-CoV-2 antibodies in the sample to form immune complexes. The immune complexes move forward along the cassette by chromatography. The IgM in the immune complexes will be captured by the pre-coated anti-human IgM on the test zone, condense the color to form the reaction line M.
The IgG in the immune complexes will be captured by the pre-coated anti-human IgG on the test zone, condense the color to form the reaction line G. The colloidal gold-labeled chicken IgY antibodies are combined with the pre-coated goat anti-chicken IgY antibodies on the quality control line to form the quality control line C.
4.Storage :
COVID-19 IgG/IgM Rapid Test Cassette should be stored in a dark place at 2-30°C for 12 months from the date of manufacture. Keep the test cassette in a sealed pouch until use. Once you have taken the test cassette out of the pouch, use it immediately. Do not use the test beyond the indicated expiration date.
5.Procedure of NewScen COVID-19 IgG/IgM Rapid Test Cassette:
5.1. Sample preparation: Fresh serum, plasma, or whole blood samples, no pretreatment is required. If the samples are stored at 2~8°C, the samples should be restored at room temperature for 15~30 minutes before use, returned to room temperature, and thoroughly mixed before testing.
5.2. Reagent preparation: Open the package, the pouch should be sealed well. If the test reagent stores in the refrigerator, it should be restored to room temperature. Then open the packaging pouch and take out the test reagent, place it on the platform.
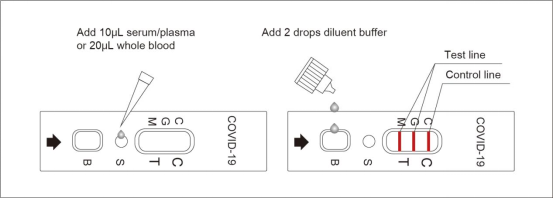
5.3. Detection and interpretation: Add 10μL serum/plasma or 20μL whole blood sample into S well, after the sample has permeated completely, add 2 drops of diluent buffer into B well. Read the result in 15~20 minutes, interpret the test result after 20 minutes may cause a false result.
6.Result Interpretation:
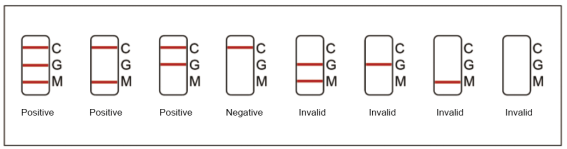
6.1. Positive of IgG and IgM: One color line in the control zone (C), one color line in the test zone (G), and one color line in the test zone (M). Indicates IgG and IgM test result is positive.
6.2. Positive of IgG: One color line in the control zone (C) and one color line in the test zone (G), indicates the IgG test result is positive.
6.3. Positive of IgM: One color line in the control zone (C) and one color line in the test zone (M), indicates the IgM test result is positive.
6.4. Negative of IgG and IgM: Only one color line in the control zone (C) indicates IgG and IgM test result is negative.
6.5. Invalid: If no color line appears in the control zone (C), the test is invalid. Discard the test cassette and perform with a new cassette.
Built-In Control:
COVID-19 IgG/IgM Rapid Test Cassette has a built-in procedural control that demonstrates assay validity. A color line appeared on the control zone (C) indicates that the test runs correctly.
7.Limitation:
7.1. The kit is only used to detect human serum, plasma, and whole blood samples.
7.2. The accuracy of the test depends on the process of sample collection. Improper sample collection, improper sample storage, or repeated freezing and thawing of samples will affect the test results.
7.3. The test results of this reagent are for clinical reference only and should not be used as the sole basis for clinical diagnosis and treatment. The clinical management of the patient should be considered in combination with other laboratory tests of the patient’s symptoms/signs history and treatment response.
7.4. Due to the limitation of antibody detection reagent methodology, nucleic acid detection or virus culture identification method is recommended to confirm the negative test results.
7.5. Too low levels of the antibodies in the sample can lead to false-negative results.
7.6. The mutation of the virus gene may change the epitopes of antibodies and result in false-negative results.
8.Sensitivity and Specificity of NewScen COVID-19 IgG/IgM Rapid Test Cassette:
9.CE Qualified:
10.International Certification of NewScen COVID-19 IgG/IgMRapid Test Cassette: