Key Words: NewScen H. Pylori Antigen Stool test Kit (Colloidal Gold)
Sample type: Stool
Detection type&Method: Qualitative Colloidal Gold
Usage/Application: Laboratory, Hospital&Private Clinic, Pathology, Community Center
Certificate: ISO9001/ ISO13485/CE/NMPA
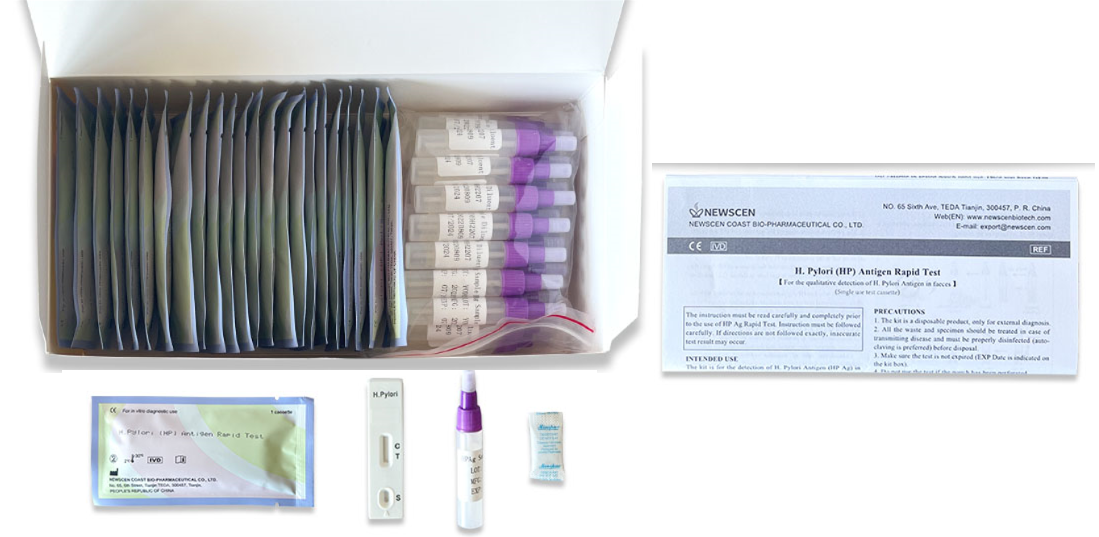
Format: Cassette, Strip, Uncut Sheet
Sensitivity: 93.9%
Specificity: 97.3%
Total Accuracy: 96.4%
Reading time: 15-30 minutes
Packaging Details:
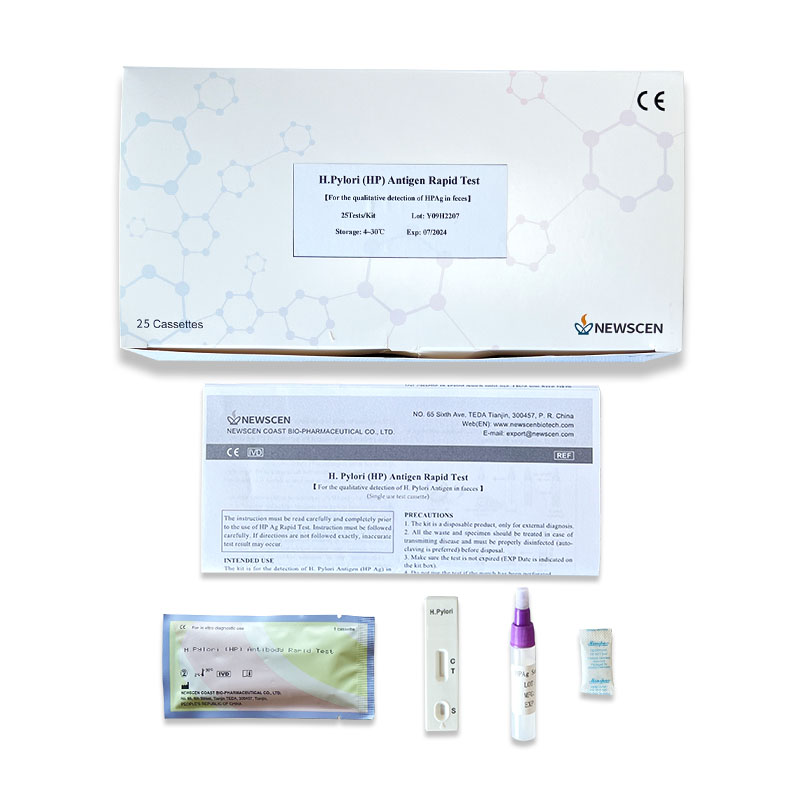
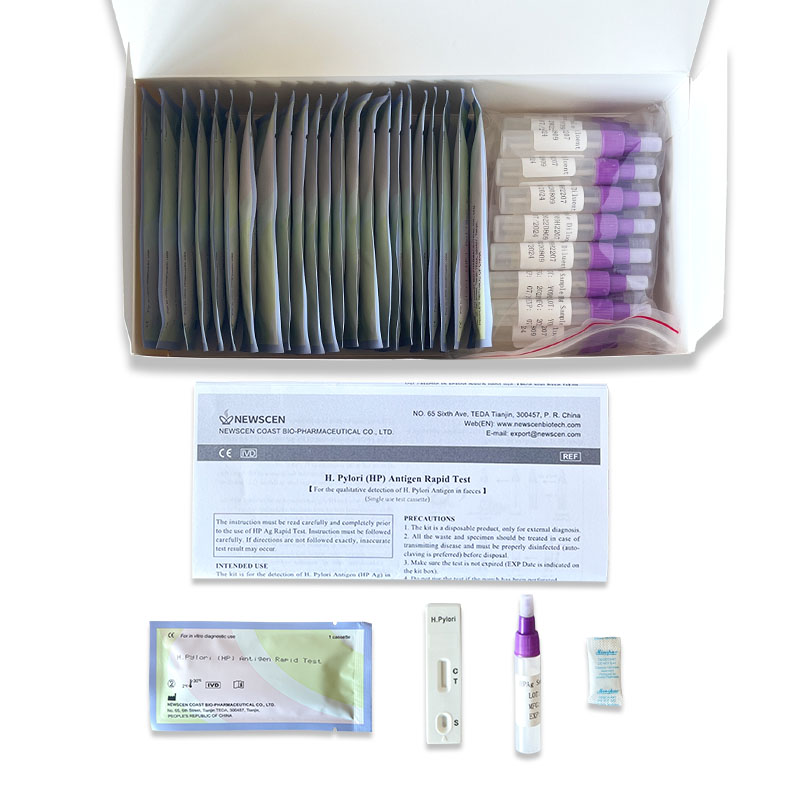
①For Professional Use: 25 Cassette/Kit; 100 Strips/Kit; 50 Strips/Bottle (For Customization)…
②Uncut Sheet for OEM
Description
H. Pylori Antigen Rapid Test Kit (Colloidal Gold) for the qualitative detection of H. Pylori Antigen in faeces.
1. Product Description:
The NewScen H. pylori Antigen Rapid Test is an in vitro qualitative immunochromatographic assay for the rapid detection of Helicobacter pylori antigens in human stool specimens. The test results are intended to aid in the diagnosis of H. pylori infection, to monitor the effectiveness of therapeutic treatment and to confirm the eradication of H. pylori in peptic ulcer patients.
The NewScen H. Pylori Antigen Rapid Test Kit is for the detection of H. Pylori Antigen (HPAg) in human faeces.
2. NewScen Provide HPAg Rapid Test With Below Specification.
3. Which conditions does Newscen HPAb Rapid Test screen for?
4. How do you test for HPAg Rapid Test Kit?
Click here to download: Instruction of Newscen HP-Ag Test Kit (25 cassettes)