Key Words: Coronavirus (SARS-CoV2) diagnostic with microfluidics chip, SARS-CoV-2 Rapid Antigen Test, Microfluidic device for COVID-19 Antigen, SARS COV 2 Variant Test
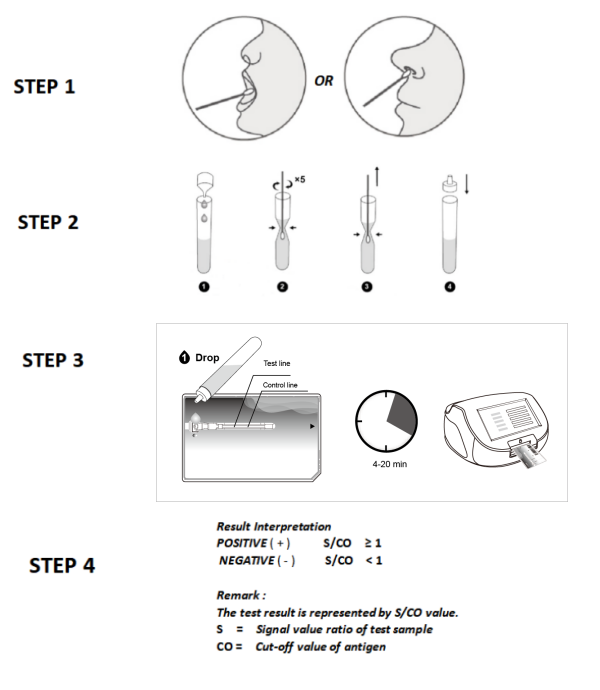
Sample type: Throat OR nasal swab
Detection type: Qualitative
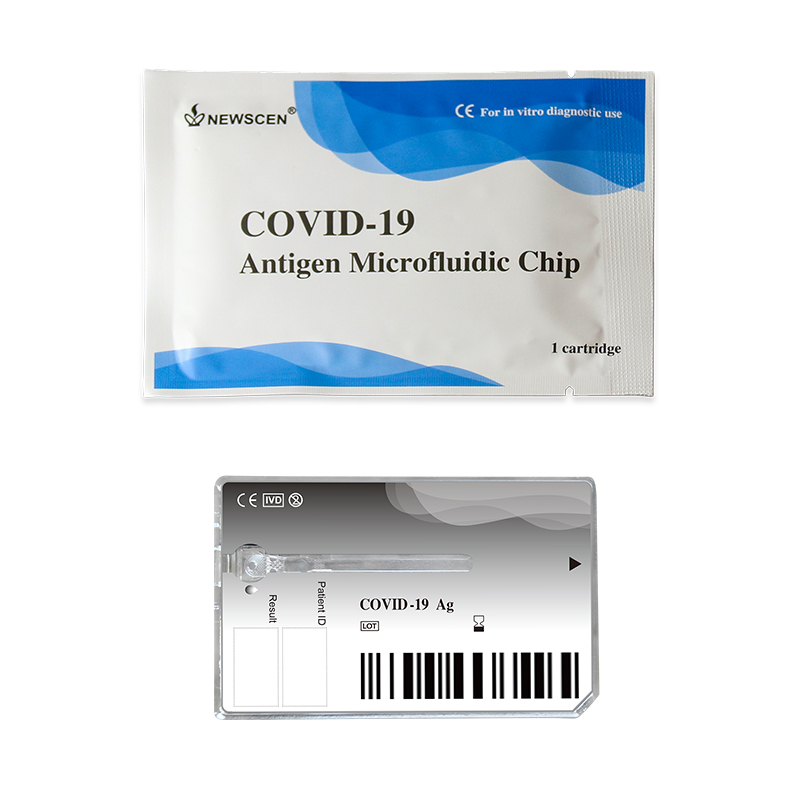
Method: Microfluidic
Function: Diagnose
Certificate: ISO9001/ ISO13485/CE
Reading time: 4 minutes
Sensitivity: 98.11%
Specificity: 99.56%
Accuracy: 99.28%
Features:
* Lab-on-a-chip technology
* CV ≤ 5%
Packaging Details:
Pouch+Box+Carton packaging
(1) With our company’s Logo
(2) With the natural package
(3) With OEM package
(4) ODM
Description
1. Product Description:
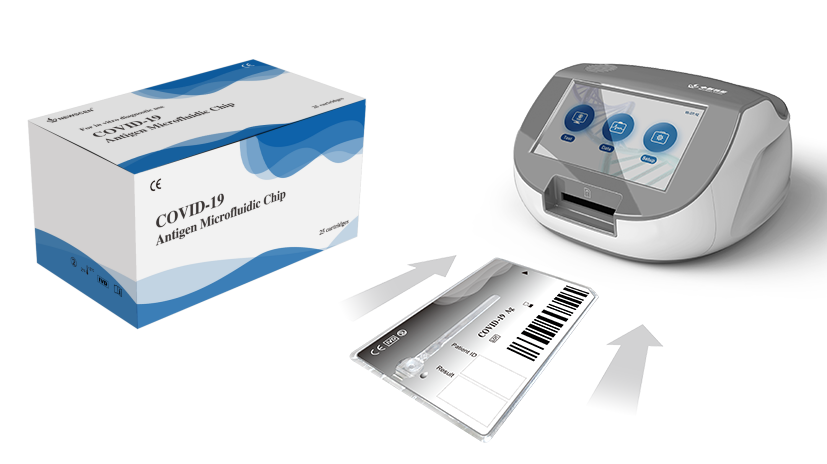
NewScen COVID-19 Antigen Microfluidic Chip is suitable for large-scale screening in multiple scenarios ( feasible in low-resource settings, P3 Lab, etc. ). For the qualitative detection of SARS-CoV-2 antigen in human throat swab or nasal swab.
It is an ideal alternative to RT-PCR for cases with suspected symptoms of COVID-19 within 7 days.
2. Intended Use:
COVID-19 Antigen Microfluidic Chip is for in vitro qualitative detection of specific antigens to SARS-CoV-2 present in the human throat or nasal cavity. It cannot be used as the basis for the diagnosis and exclusion of COVID-19.
The kit is used in combination with F10Pro NewScen Fluorimetric Immunoassay Analyser produced by our company. This reagent is used to detect cases with suspected symptoms of COVID-19 within 7 days.
If suspected symptoms are more than 7 days, it is recommended to test with COVID-19 antibodies or nucleic acid reagents. The main clinical symptoms of COVID-19 are: fever, dry cough, fatigue, a few patients will have a stuffy nose, runny nose, and diarrhea.
3.Principle:
This kit uses microfluidic technology, fluorescence technology, and double antibody sandwich technique of comprehensive detection technology to detect specific antigens to SARS-CoV-2 in the human throat or nasal cavity. Microfluidic technology is a detection technology that uses the microchannel inside the reagent cartridge to realize liquid quantification and uniform flow.
During the detection, SARS-CoV-2 antigen in the sample first react with the fluorescently labeled specific antibody to form the antigen~fluorescently labeled antibody immune complexes, the immune complexes depend on siphon action to flow forward automatically in the microchannel. When flow to the detection area, meet coated paired antibodies and be absorbed in the detection area.
The unbound fluorescent-labeled antibodies continue to flow forward, and when they flow to the quality control area, encounter the SARS-CoV-2 recombinant antigens, and absorbed in the quality control area, the residuum eventually flows to the waste area.
On the NewScen Fluorimetric Immunoassay Analyser, it collects the fluorescence signal that excited by the excitation light in the detection area and the quality control area, the intensity of the fluorescence signal is positively correlated with the amount of SARS-CoV-2 antigen present in the specimen.
4. Storage:
COVID-19 Antigen Microfluidic Chip should be stored in a dark place at 2~8°C for 12 months from the date of manufacture. Keep the test cartridge in a sealed pouch until use. Once you have taken the test cartridge out of the pouch, use it immediately. Do not use the test beyond the indicated expiration date.
5. Procedure of NewScen COVID-19 Antigen Microfluidic Chip: https://youtu.be/2zbMzytcMys
6.Limitation:
6.1. The kit is only used to detect human throat swabs or nasal swabs.
6.2. The accuracy of the test depends on the process of sample collection. Improper sample collection, improper sample storage, or repeated freezing and thawing of samples will affect the test results.
6.3. The test results of this reagent are for clinical reference only and should not be used as the sole basis for clinical diagnosis and treatment. The clinical management of the patient should be considered in combination with other laboratory tests of the patient’s symptoms/signs history and treatment response.
7.CE Qualified: